At least four Russian military satellites changed their orbits to match that of a Finnish-American radar surveillance satellite in the last week, raising questions about Russia's intentions amid an ever-expanding standoff high above Earth.
The maneuvers were identified through open source orbital tracking data. Greg Gillinger, a retired Air Force space intelligence officer, revealed the orbit changes Friday in a special edition of his Integrity Flash newsletter, published by Integrity ISR, a private business that provides "combat-proven operational support and elite training that enhances mission success across ISR (Intelligence, Surveillance, and Reconnaissance), cyber, space, and targeting domains."
The Russian satellites in question, designated Kosmos 2610 through 2613, launched together on April 16 on a Soyuz-2.1b rocket from Plesetsk Cosmodrome in northern Russia. Over the last week or so, the four satellites adjusted their inclinations—the angles of their orbits to the equator—by less than a degree.
That may sound insignificant, but such "plane change" maneuvers use up a lot of fuel. The delta-v, or velocity change, required for a plane change maneuver of this magnitude is equivalent to the impulse needed to raise altitude by more than 100 miles.
The upshot is that these four Russian satellites are now positioned to routinely pass near a commercial radar surveillance satellite operated by the Finnish-American company ICEYE. This imaging platform, named ICEYE-X36, is part of a fleet of satellites providing all-weather overhead radar images to the US military and European governments. ICEYE also provides imagery to Ukraine's military in its fight against Russia. ICEYE's co-founder and CEO, Rafal Modrzewski, met with Ukrainian President Volodymyr Zelenskyy last year.
According to Gillinger, the cross-track distances between the four Russian satellites and ICEYE-X36 now range between about 500 meters (1,640 feet) and 22 kilometers (13.7 miles). All of this is taking place in polar orbit at an altitude of approximately 340 miles (547 kilometers).
Russian satellite operators are now in a position to close in on the ICEYE satellite with "minor adjustments" in "satellite eccentricity and average altitudes," Gillinger wrote in his newsletter. A fifth satellite from the same Russian launch last month now appears to be performing a similar set of maneuvers to move closer to ICEYE-X36.
Co-planar or cosplay?
We know little about what these particular Kosmos satellites can do. Perhaps, as one retired US military space official recently told Ars, this is another example of Russia rattling a dull saber. Russian military officials seem to enjoy probing US and allied forces, often flying strategic bombers near US and European airspace.
This same behavior now appears to extend into space, with Russia's launch of several military spacecraft shadowing the US government's most sophisticated spy satellites in low-Earth orbit several hundred miles above the planet. US officials believe at least some of these Russian satellites are part of an anti-satellite weapons program.
More recently, a mysterious Russian military satellite arrived in geosynchronous orbit more than 22,000 miles over the equator. Circumstantial evidence suggests this, too, may be part of a Russian anti-satellite system. The US Space Force dispatched one of its own inspection satellites in geosynchronous orbit to get a closer look.
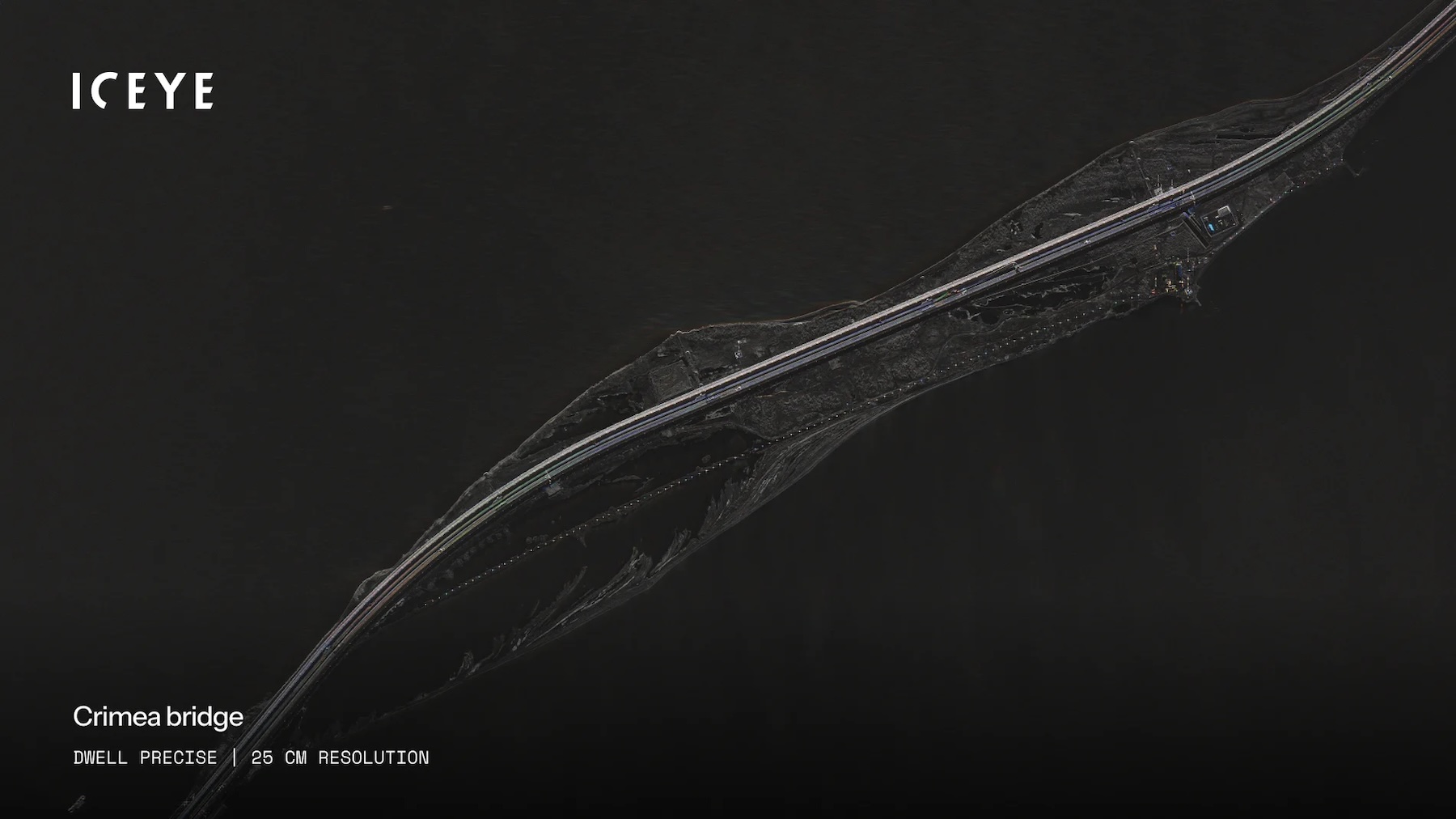
A radar image of a bridge in Crimea taken by an ICEYE satellite.
Credit:
ICEYE
Targeting a single spacecraft, such as ICEYE-X36, in a constellation of similar imaging satellites would do little to inhibit the access of Ukraine or other Western nations to radar surveillance imagery. ICEYE, itself, operates dozens more radar imaging satellites. Unlike optical spy satellites, radars provide imagery day and night, regardless of cloud cover.
But Russia's maneuvers to match the plane of ICEYE-X36's orbit appear to be intentional. Russian military satellites have conducted similar operations to move into "co-planar" orbits with Keyhole-class spy satellites owned by the National Reconnaissance Office. The recent maneuvers with Kosmos 2610 and its cohorts appear to mimic what Russia has done to move within striking distance of the NRO's satellites.
"We do not know Russia's intentions or the capabilities of these particular satellites," Gillinger wrote. "However, maneuvering into a co-planar orbit is alarming. Plane matching is the first (and most fuel expensive) step to conducting Rendezvous Proximity Operations (RPO), likely necessary for the Russian satellites to target (kinetically or non-kinetically) ICEYE-X36.
"We also do not know the satellites' total fuel capacity, however the expenditure [during these maneuvers] is evidence the satellites are capable of conducting high-energy maneuvers," Gillinger continued. "This capability is not common for satellites conducting typical Earth observation, signal collection, or communications missions."
ICEYE did not respond to questions from Ars on Friday; the company announced in January that it was expanding its partnership with the Ukrainian military. ICEYE's newest satellite capture images with a resolution of up to 16 centimeters, about the size of a grapefruit.
"ICEYE is proud and humbled to have supported Ukraine's defense teams with reliable, near-real-time space-based intelligence since the beginning of the invasion," said John Cartwright, senior vice president of data product at ICEYE.
"This agreement strengthens assured access to our high-resolution SAR imagery, helping Ukraine make decisions faster and with greater confidence," Cartwright said. "ICEYE stands firmly with Ukraine in the face of these hostilities, and is deeply committed to strengthening Europe's security more widely by ensuring our allies have the best decision-ready data when they need it most."
Read full article
Comments


 A radar image of a bridge in Crimea taken by an ICEYE satellite.
Credit:
ICEYE
A radar image of a bridge in Crimea taken by an ICEYE satellite.
Credit:
ICEYE

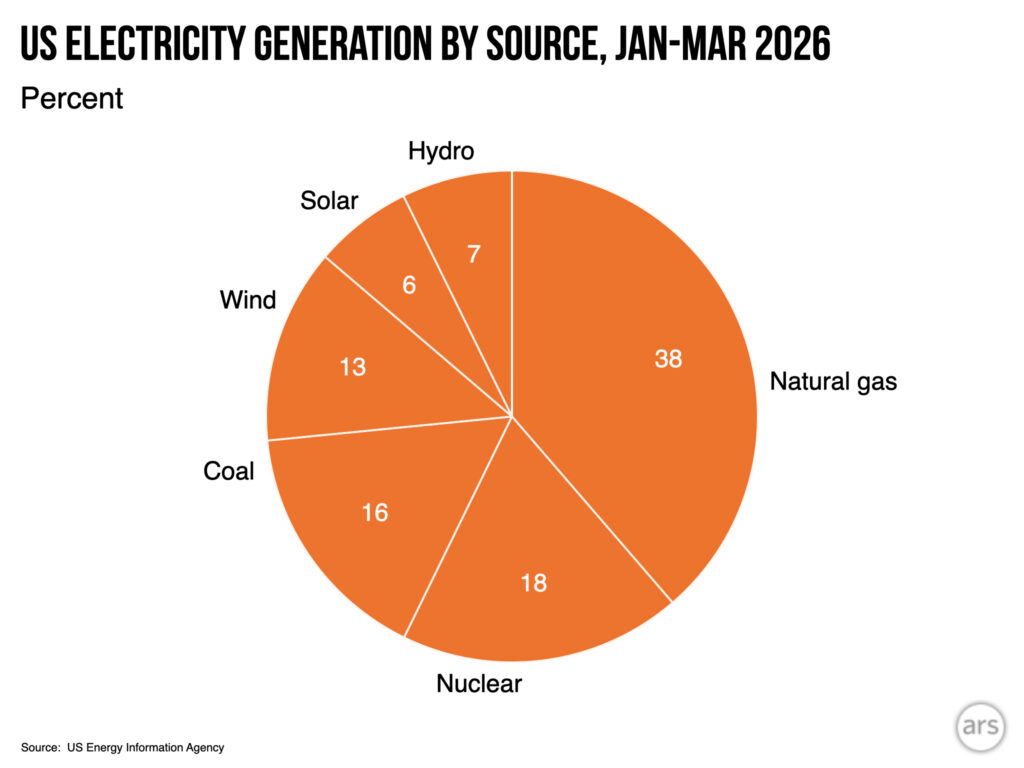
 Wind and hydro are up, coal is down compared to this period a year earlier.
Credit:
John Timmer
Wind and hydro are up, coal is down compared to this period a year earlier.
Credit:
John Timmer